Build Your Knowledge About Protein and Amino Acids
All biological organisms—including humans—are made of protein. The human body alone contains an estimated two million different proteins. Across all living land organisms, that number likely exceeds ten million. Proteins are essential for providing both structure and function in the body, supporting everything from muscles and organs to chemical reactions and communication between cells. To maintain balance, the body continually produces specialized proteins that build, regulate, and break down other proteins. Understanding how your body uses protein—and how to supply it through your diet—is key to supporting overall health.
Protein Structure
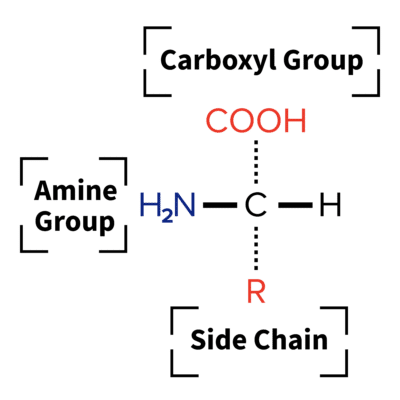
Proteins are long-chained molecules made from amino acids, the basic building blocks of life. Amino Acids are organic compounds that have a very specific chemical structure:
- Amine: molecules with a nitrogen and two hydrogen atoms
- Carboxyl: molecules containing carbon, two oxygen, and one hydrogen molecule
- Side chain (R group): molecules specific to each amino acid.
There are 20 well-known amino acids that make up the proteins in your body. In addition to the 20 standard amino acids, two others—selenocysteine and pyrrolysine—have been identified as protein‑building amino acids. All animal and plant proteins on earth are constructed from these 22 amino acids.
In 1806, French chemists Louis-Nicolas Vauquelin and Pierre Jean Robiquet isolated a compound in asparagus that was subsequently named asparagine. It was the first amino acid discovered.
Even with a somewhat limited number of amino acids, proteins can be very complex molecules. That’s because most proteins fold into unique, three-dimensional structures.
Biochemists often refer to four distinct aspects of a protein’s structure:
- Primary structure: The basic order of amino acids in a protein, linked together by peptide bonds. Short chains of amino acids are called peptides, and they usually have fewer than 50 amino acids. The smallest peptides, called dipeptides, contain just two amino acids.
- Secondary structure: The three-dimensional form of local or specific segments of proteins. These local segments are twists and turns stabilized by hydrogen bonds.
- Tertiary structure: The overall shape of a single protein molecule. Also synonymous with the term “fold,” the tertiary structure controls the basic function of the protein.
- Quaternary structure: The arrangement formed when two or more protein chains come together to work as one functional protein. Each individual chain is called a subunit. These chains, known as polypeptides, are long, unbroken strands of amino acids.
The longest known protein, titin, is also known as connectin. It contains 26,926 amino acids. Titin is found in muscle and contributes to its passive stiffness.
Protein Function
Proteins serve many vital functions in the body. They regulate bodily systems and structural components that give cells, tissues, and organs their shape and strength. Proteins also play key roles in cell communication, immune defense, nutrient transport, and tissue repair. Because of their extensive responsibilities, proteins are constantly being made, used, and recycled to support growth, maintenance, and overall health.
Enzymes and Hormones
The best-known role of proteins in the cell is as enzymes, which catalyze chemical reactions. For example, digestive enzymes help metabolize your food into cellular energy. Enzymes can also take the form of antioxidants. Selenium-containing protein glutathione peroxidase is one of the most important antioxidant compounds in your body.
Certain hormones are proteins made from amino acids. This includes sex hormones known as gonadotropins, as well as endocrine hormones like insulin and glucagon, which help regulate processes such as reproduction, metabolism, and blood sugar control.
Cell signaling
Many proteins are involved in the process of cell signaling and signal transduction. Some proteins, such as insulin, transmit a signal from the cell in which they were synthesized to other cells in distant tissues.
In 1955, insulin became the first protein to be fully sequenced. British biochemist Frederick Sanger received the Nobel Prize in Chemistry for this groundbreaking research.
Proteins participate in both sides of the signaling process. They also act as receptors on the cells that bind to a signaling molecule to induce a biochemical response.
For example, antibodies are proteins that play an important role in immune cell signaling. When an antibody binds to a foreign substance (called an antigen), it acts as a signal that alerts other immune cells to the presence of a threat. This binding event triggers a cascade of cellular responses—such as activating immune cells, releasing signaling molecules, or marking the invader for destruction—allowing the immune system to coordinate an effective defense.
Recombinant DNA techniques were used in 1978 by biotechnology firm Genentech to produce synthetic “human” insulin, making insulin the first human protein to be manufactured through biotechnology.
Structural proteins
Structural proteins provide shape and rigidity to biological components. Most structural proteins are fibrous in nature.
Collagen and elastin are two examples of these tough, fibrous proteins. They’re both critical components of connective tissue, like cartilage. They serve major functions in the structure of skin.
The protein keratin provides structure for your hair and nails. In the animal kingdom, keratin helps make feathers, hooves, and some animal shells.
There are also globular proteins that form long, stiff fibers that allow cells to maintain their shape and size.
Other functions
Proteins also play roles in cell adhesion, growth factors, nutrient transport and storage, and much more. In humans, non-protein amino acids also have important roles as neurotransmitters or precursors to other molecules. Neurotransmitters are substances that help transfer impulses between nerves.
- Tryptophan is a precursor of the neurotransmitter serotonin, which is involved in mood and much more.
- Tyrosine (and its precursor phenylalanine) are precursors of dopamine, epinephrine, and norepinephrine and various trace amines.
- Glycine is a precursor of important compounds in the blood known as porphyrins (i.e. heme).
- Arginine is a precursor of nitric oxide.
Protein and Amino Acids in the Diet
You don’t eat a chicken breast and add that exact same type of protein to your body. This is because the proteins you eat are not absorbed intact. Proteins are digested into amino acids or small peptides that can be absorbed by the intestine and transported in the blood.
Protein digestion begins in the stomach. The acidic environment and the stomach enzyme pepsin begin to denature (unravel) the protein. This makes it more accessible to digestive enzymes secreted by the pancreas.
Other specific enzymes further digest the larger peptides into di- and tripeptides and free amino acids. These are then released into the blood for use by other tissues. After digestion, the amino acids are used to synthesize proteins and other biomolecules or they’re converted to urea and carbon dioxide as a source of energy.
Of the 20 standard amino acids, nine are called essential because your body cannot make them from other compounds at the level you need for normal growth. They must be obtained from food. The essential amino acids include histidine, isoleucine, leucine, lysine, methionine, phenylalanine, threonine, tryptophan, and valine.
Protein Quality
Not all dietary proteins are equally effective for your health. Scientists have developed a scale to let you know a protein’s quality. Previous methods of measuring quality include the PER (protein efficiency ratio) and BV (biological value). But they’re rarely used today.
One method of determining protein quality is known as protein digestibility-corrected amino acid score (PDCAAS). The PDCAAS allows evaluation of food protein quality based on your needs. It measures the quality of a protein based on the amino acid requirements (adjusted for digestibility) of a 2- to 5-year-old child (considered the most nutritionally demanding age group).
Using the PDCAAS method, the protein quality rankings are determined by comparing the amino acid profile of the specific food protein against a standard amino acid profile. The highest possible score is a 1.0. This score means, after digestion of the protein, it provides 100 percent or more of the essential amino acids required—per unit of protein.
The current recommended method for measuring protein quality is the digestible indispensable amino acid score (DIAAS). Rather than looking at overall protein digestion, DIAAS focuses on how well individual essential amino acids are digested and absorbed in the small intestine. This provides a more accurate picture of how well a protein can meet the body’s needs.
DIAAS works by comparing the digestible amino acid content of a food to human amino acid needs. Each essential amino acid receives a score, and the lowest score determines the overall protein quality. For example, high‑quality animal proteins such as milk or eggs typically have high DIAAS values, meaning they provide all essential amino acids in amounts the body can easily use. Some plant‑based proteins may have lower DIAAS values, because one or more essential amino acids are present in smaller amounts or are less easily digested.
Protein Requirements
Daily protein needs are combinations of many factors, including age, body composition, gender, total energy intake (calories), training status, and level of exercise for an athlete.
The recommendation for protein intake is based on body weight. For normally healthy adults the daily recommendation is 0.8 grams of protein for every kilogram of body weight. For a normally healthy adult woman who weighs 150 pounds and is sedentary, that translates to about 55 grams of protein a day.
The recommended dietary allowance (RDA) for adult females is 46 grams/day, for adult men it is 56 grams/day, and for pregnant and lactating women it is 71 grams/day. That doesn’t match up with the recommendations for a reason.
Much like the RDAs for micronutrients, they’re meant to provide sufficient levels to allow for normal health and growth in apparently healthy individuals. The RDA is the minimum level of intake needed to meet basic nutritional requirements, but it isn’t necessarily the specific amount that is best or optimal.
Protein for Athletes
Athletes consume dietary protein to repair and rebuild skeletal muscle and connective tissues following intense training bouts or athletic events. Athletes should generally get at least 1.4 to 2.0 grams of protein for every kilogram of body weight per day.
One specific type of amino acids is especially important for athletes— branched-chain amino acids (BCAAs). Of the nine essential amino acids, only three are known as BCAAs. But they make up about 35 percent of the proteins in muscle tissue. These BCAAs are also known to play a beneficial role during aerobic exercise by decreasing the rate of protein degradation and delaying muscle glycogen depletion.
The three BCAAs are leucine, isoleucine, and valine. BCAAs are crucial to protein synthesis and turnover, cell signaling pathways, and glucose metabolism. They also play roles in the synthesis of proteins and neurotransmitters in the brain, and the production of energy.
A growing amount of scientific literature suggests that of the three BCAAs, leucine appears to play the most significant role in stimulating protein synthesis.
Protein for the Elderly
Normal aging is associated with changes in muscle protein metabolism and decreased response to amino acids in a meal. Adequate leucine intake during a meal may help overcome this insensitivity to muscle proteins and be a useful tool in the prevention of excess muscle loss. The minimum dosage for these benefits is likely between 2.0 and 2.5 grams of leucine per serving or meal.
It is also becoming clear that leucine taken as part of a whole quality protein is likely more beneficial than supplementing with BCAAs alone. To maximize muscle protein retention in the elderly and provide adequate leucine, most experts propose dietary plans that include 25-30 grams of high-quality protein per meal.
Muscle loss occurs with age but losing more than normal (0.5-1.0%/year) is known as sarcopenia. It’s a progressive decline in skeletal muscle mass which leads to decreased strength and functionality. Sarcopenia occurs in as much as 30% of individuals over 60 years of age and more than 50% of those over 80 years.
Vegans and Vegetarians
Plant proteins by themselves can provide enough quality protein to sustain healthy growth, repair, and muscle function. However, many plant proteins provide less than optimal levels of all essential amino acids. For vegans or vegetarians, it simply becomes more important to get a wide variety of foods that include beans, legumes, nuts, and seeds. Typically, a vegetarian diet that is highly varied and adequate in calories will provide plenty of quality protein.
Overall Recommendation
Based on the totality of the available research, a safe and healthy range of protein to aim for is roughly 15-25 percent of total dietary calories—with some adjustments for activity level and age. For an average 2,000 calorie intake, this would represent an intake of 75-125 grams of protein per day.
Food Sources—Choosing the Right Protein
Just like fat and carbohydrates, protein is a macronutrient. Unlike fat and carbohydrates, usable protein is not stored in significant amounts. Your amino acid stores are constantly used and must be replenished often. If you aren’t getting enough protein in your diet, your body will take it from your muscle mass.
There is no single best protein source. While there are certainly some foods that would be considered better protein sources, a varied and mixed diet typically provides adequate protein intake: seafood, lean poultry, beans, eggs, dairy (yogurt, milk, and cheese), soy and other legumes, lean beef, and pork. Quinoa, pumpkin seeds, and pistachios are also excellent sources of protein. Focusing on quality whole foods will help you achieve an adequate intake of important nutrients and proteins.
If you are having a hard time meeting your protein needs or don’t have the time for a sit-down meal, a quality meal-replacement drink or bar can be a beneficial addition to your protein intake for the day.
More Protein Facts
- The word protein is derived from the Greek proteios “the first quality,” or from protos “first.”
- As long as you are healthy and well-hydrated, high protein intakes do not damage healthy kidneys.
- The terms “complete” and “incomplete” are misleading in relation to plant protein. Getting a variety of protein from plants supplies enough of all essential amino acids when caloric requirements are met.
- Protein, like carbohydrates, supplies approximately four calories of energy per gram. Fat provides nine calories per gram, and alcohol provides seven calories per gram.
- Protein is first utilized for growth and repair. Excess can be used for energy. Protein not needed for these functions will be converted and stored as fat.
- Carbohydrates are not the only stimulus for insulin secretion. Protein can also stimulate insulin secretion, but to a lesser degree and without raising blood glucose levels.
- Protein is more satiating than fat or carbohydrates. Some research shows that a modest increase in protein content and a modest reduction in glycemic index can lead to an improvement in compliance and maintenance of a healthy weight.
References
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4889822/
https://www.ncbi.nlm.nih.gov/books/NBK22600/
https://jissn.biomedcentral.com/articles/10.1186/s12970-017-0177-8
https://www.ncbi.nlm.nih.gov/books/NBK56068/table/summarytables.t4/?report=objectonly
https://www.ncbi.nlm.nih.gov/pubmed/16365098
http://www.theissnscoop.com/tag/bcaa/
https://jissn.biomedcentral.com/articles/10.1186/1550-2783-7-7
http://journals.sagepub.com/doi/abs/10.1177/0115426506021005430
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2760315/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3359496/
https://askthescientists.com/collagen/
https://askthescientists.com/eating-bugs/
https://askthescientists.com/milk-types/